 The addition of ethylene oxide increases water solubility, propylene oxide decreases it.Ĭommercially available demulsifier formulations are typically a mixture of two to four different chemistries, in carrier solvent(s) such as xylene, heavy aromatic naphtha (HAN), Isopropanol, methanol, 2-Ethylhexanol or diesel. A colloid of two immiscible liquids in which one fluid contains a dispersion of the other fluid is known as an emulsion. Examples of simple emulsions are water in oil emulsion. 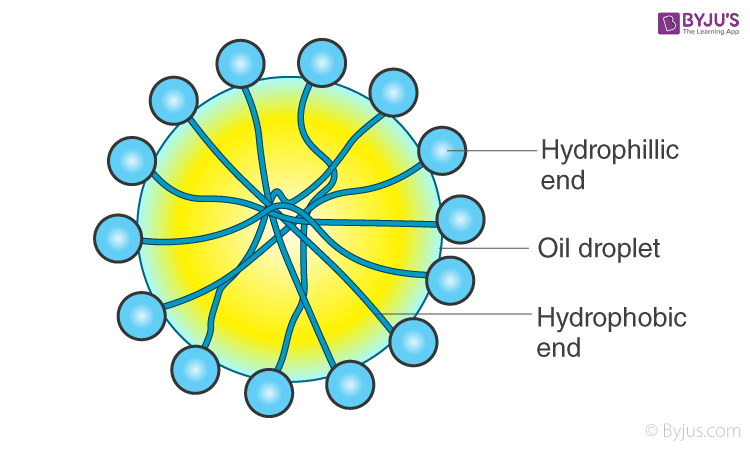
A number of emulsifiers are derived from algae, among them algin, carrageenan, and agar. The above are usually ethoxylated (and/or propoxylated) to provide the desired degree of water/oil solubility. Emulsions formed by mixing oil in water, water in oil, etc., are known as simple emulsions. emulsifier, in foods, any of numerous chemical additives that encourage the suspension of one liquid in another, as in the mixture of oil and water in margarine, shortening, ice cream, and salad dressing. Base catalysed phenol-formaldehyde resins.Acid catalysed phenol-formaldehyde resins.Emulsions are part of a more general class of two-phase systems of matter called colloids. If the majority of the water and salt are not removed, significant corrosion problems can occur in the refining process.ĭemulsifiers are typically based on the following chemistry: An emulsion is a mixture of two or more liquids that are normally immiscible (unmixable or unblendable) owing to liquid-liquid phase separation. Demulsifiers, or emulsion breakers, are a class of specialty chemicals used to separate emulsions, for example, water in oil. This water (and salt) must be removed from the crude oil prior to refining. 
They are commonly used in the processing of crude oil, which is typically produced along with significant quantities of saline water. 1 As such, when two entirely aqueous solutions containing different water-soluble molecules are mixed, water. Demulsifiers, or emulsion breakers, are a class of specialty chemicals used to separate emulsions, for example, water in oil. Water-in-water (W/W) emulsion is a system that consists of droplets of water-solvated molecules in another continuous aqueous solution both the droplet and continuous phases contain different molecules that are entirely water-soluble. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
